You must be logged in to post a review.
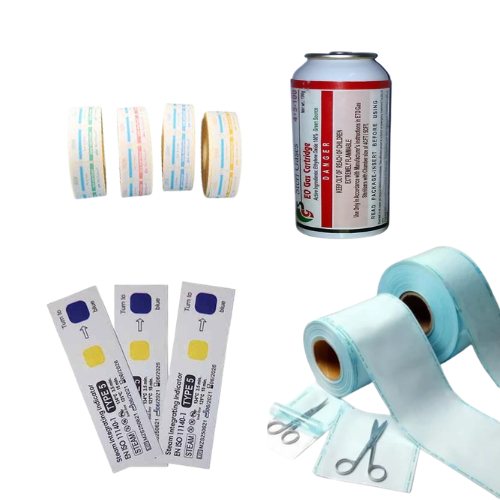
CSSD Consumables
-
Sterility assurance – packaged and processed to maintain sterility until point-of-use (validated barrier systems, peel pouches, sterility indicators).
-
Biocompatibility & safety – materials that won’t leach toxins or cause reactions; safe for contact with instruments and patients.
-
Durability & performance – withstand sterilization cycles (autoclave, EO, H₂O₂) without degrading or losing function.
-
Traceability & labeling – clear batch/lot, expiry, and sterilization indicator info to support inventory control and recall/QA processes.
-
Regulatory compliance & quality control – manufactured and tested to relevant standards (ISO, local health regs) with lot testing and documentation.





Reviews
There are no reviews yet.